
Cynthia X. Ma, MD, PhD
Professor of Medicine
- Phone: 314-747-1171
- Fax: 314-362-7086
- Email: cynthiaxma@nospam.wustl.edu
Address:
Division of Oncology
Mail Stop 8076-0041-03
Washington University
660 South Euclid Avenue
St. Louis, MO 63110
Admin:
Janet Weier
jweier@wustl.edu
- Breast cancer
- Pharmacogenomics of anticancer agents
- Predictive biomarkers
- Developmental therapeutics
Dr. Ma is a physician scientist with a research focus on pre-clinical and early phase clinical investigations in developing molecular therapeutics for breast cancer through collaborative research. Dr. Ma has designed and conducted several investigator-initiated trials in genomic biomarker focused breast cancer populations, including the neoadjuvant AKT inhibitor trial in the PIK3CA mutant population, the neoadjuvant CDK4/6 inhibitor in the PIK3CA mutant and WT population, as well as the trial of neratinib in metastatic HER2 non-amplified but mutant patient population. She is also leading the phase III Alliance trial A011106 to validate neoadjuvant biomarkers predictive of long-term outcomes and to investigate endocrine resistance mechanisms for estrogen receptor positive breast cancer. In collaboration with NCI CTEP, Dr. Ma designed a phase I/II trial testing whether the addition of PI3K inhibitor copanlisib to standard of care CDK4/6 inhibitor abemaciclib and fulvestrant in patients with endocrine resistance disease, which will be activated in the last quarter of 2019.
In addition to clinical trial development, the Ma laboratory studies clinical specimens and patient-derived xenograft models to investigate biomarkers of responsiveness for targeted agents. One particular focus in the evaluation of the role of phosphatidylinositol 3-kinase (PI3K) pathway inhibitors in the treatment of triple negative breast cancer (TNBC: negative for the expression of estrogen receptor, progesterone receptor and HER2 gene amplification) and in overcoming CDK4/6 inhibitor resistance in estrogen receptor positive breast cancer.
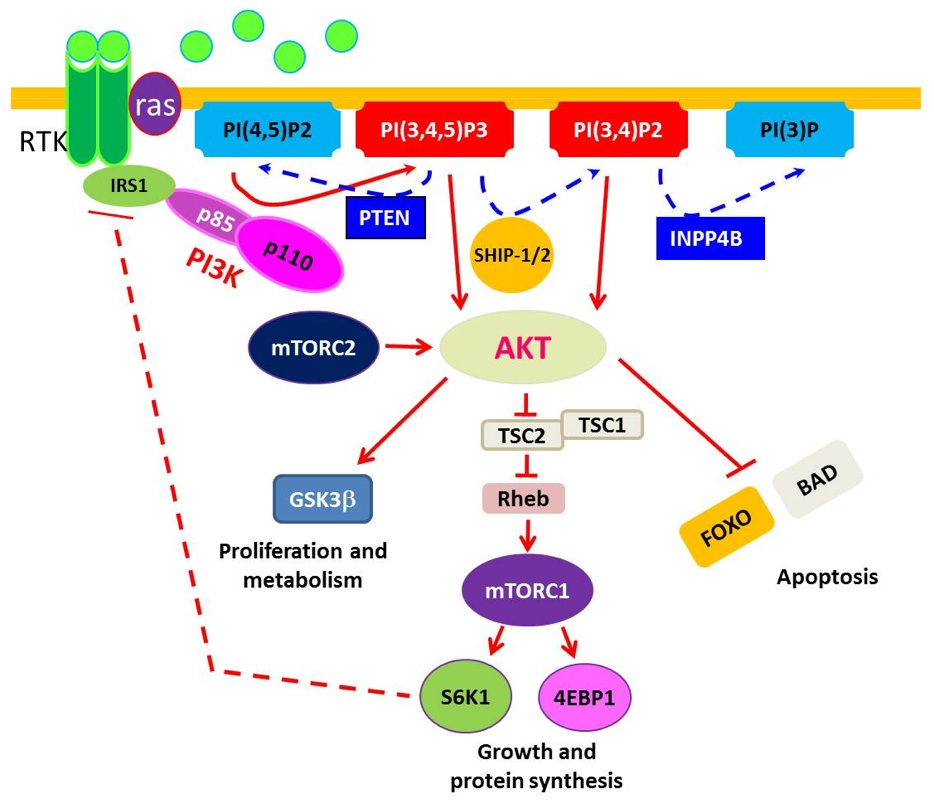
PI3K pathway signaling plays key regulatory roles in many cellular processes, including cell survival, proliferation, differentiation and angiogenesis (see Figure). PIK3CA mutation occurs in approximately 40% of estrogen receptor positive breast cancer. Although mutations in PIK3CA is 7-9% in TNBC, hyperactivation of the PI3K/AKT pathway is common, often due to loss of PTEN expression. A significantly higher level of Akt phosphorylation has been observed in TNBC patient specimens compared with non-TNBC cases. Loss of PTEN or INPP4B has been the most frequently implicated culprit for such activation in TNBC. In addition, increased levels of pAKT correlated with loss of PTEN protein and DNA copy number. The data regarding the importance of PTEN are consistent with observations of PTEN inactivation leading to “Basal-like” breast cancer in animal models. In the recent breast cancer TCGA (The Cancer Genome Atlas) report, PI3K pathway activity, either by gene expression signature or reverse phase protein array (RPPA) phosphoproteomic signature, was highest in basal-like breast cancer.
To evaluate the therapeutic potential of PI3K pathway inhibition in TNBC, we conducted a preclinical trial of PI3K inhibitor in patient-derived xenograft (PDX) models. In-depth proteomic analysis identified NEK9 and MAP2K4 as potential resistance mechanisms. Further studies are ongoing in the laboratory to investigate the signaling pathways impacted by NEK9 and MAP2K4. Additional studies are ongoing to develop rationale PI3K inhibitor combination strategies to treat TNBC and resistant estrogen receptor positive breast cancer.
Biosketch
Education
- 1997: PhD in Developmental Biology, University of Cincinnati, Cincinnati, OH
- 1990: MD, Beijing Medical University, Beijing, P. R. China
Post-Graduate Training
- 2005-2001: Hematology and Medical Oncology Fellowship, Department of Internal Medicine, Mayo Clinic, Rochester, MN
- 2001-1999: Internal Medicine Resident, New Hanover Regional Medical Center, Wilmington, NC
- 1999-1998: Internal Medicine Intern, New Hanover Regional Medical Center, Wilmington, NC
- 1998-1997: Postdoctoral Research Fellow, Division of Endocrinology, Department of Internal Medicine, University of Cincinnati, Cincinnati, OH
- 1997-1991: PhD Graduate Training, Department of Developmental Biology, University of Cincinnati, Cincinnati, OH
- 1991-1990: Internal Medicine Residency, The 1st Teaching Hospital, Beijing Medical University, Beijing, P. R. China
Academic Positions & Employment
- present-2018: Professor, Division of Oncology, Department of Medicine, Washington University, St. Louis, MO
- present-2017: Co-leader, Breast Cancer Focus Group, Siteman Cancer Center
- present-2014: Clinical Director, Breast Cancer Program, Division of Oncology, Washington University School of Medicine, St. Louis, MO
- 2018-2012: Associate Professor, Division of Oncology, Department of Medicine, Washington University, St. Louis, MO
- 2014-2010: Director, Developmental Therapeutics, Breast Oncology Section, Division of Oncology, Washington University School of Medicine, St. Louis, MO
- 2012-2005: Assistant Professor, Division of Oncology, Department of Medicine, Washington University, St. Louis, MO
University & Hospital Appointments & Committees
- present-2015: NCCN Antiemetic Guideline Committee, Siteman Cancer Center
- 2015: Committee on Admissions, Washington University School of Medicine
- 2015-2008: Human Research Protection Committee, Washington University School of Medicine
- 2016: Genetic Counseling Steering Committee, Siteman Cancer Center
Board Certification
- 2015, 2005: American Board of Internal Medicine, Medical Oncology
- 2011, 2001: Diplomate, American Board of Internal Medicine, Internal Medicine
- 2004: American Board of Internal Medicine, Hematology
Honors & Awards
- 2015-2013: NCI Cancer Clinical Investigator Team Leadership Award
- 2013-2011: CALGB Clinical Scholar Award
- 2011-2008: ASCO Career Development Award
- 2005: ASCPT Poster Presentation Blue Ribbon Award
- 2005: ASCO Merit Award
- 2005: Outstanding Fellows Achievement Award, Medical Oncology, Mayo Clinic, Rochester, MN
- 1997-1995: Albert J. Ryan Fellow, University of Cincinnati, Cincinnati, OH
National & International Committees
- present-2020: AACR Breast Cancer Research Grants Scientific Review Committee
- present-2019: NCCN Abemaciclib RFP Development Committee
- present-2019: ASCO Male Breast Cancer Consensus Guideline Panel
- present-2018: Committee of Overseas Experts (COE), Chinese Society of Clinical Oncology International
- present-2015: NCCN Antiemesis Panel
- present-2012: Breast Committee, Alliance for Clinical Trials in Oncology
- 2021-2020: NCI CTEP Ipatasertib Project Team (Translational Scientist Member)
- 2020-2017: AACR Outstanding Investigator Award in Breast Cancer Research Selection Committee
- 2017: NCI CTEP Copanlisib Project Team (Clinician Scientist Member)
- 2016-2012: Chair, Education Committee, Chinese American Hematologist and Oncologist Network
- 2015-2008: Chair, Breast Cancer Disease Group, Mayo Phase II Consortium
- 2013: AACR Annual Meeting Education Session Committee
- 2012-2009: Medical Oncology Committee, American College of Surgeons Oncology Group
- 2012-2007: Breast Committee, Cancer and Leukemia Group B
Editorial Boards
- 2020: Cancers
- present-2019: Journal of the National Cancer Institute
- present-2018: Clinical Breast Cancer
- present-2012: Breast Cancer Research and Treatment
Professional Societies
- present-2007: Chinese American Hematologist Oncologist Network
- present-2001: American Society of Clinical Oncology
- present-2001: American Association of Cancer Research
- present-1998: American Medical Association