The Division of Oncology at WashU Medicine/Siteman Cancer Center leads one of the best and most robust clinical cancer research programs in the United States. In 2022, the Division enrolled 2799 patients in more than 600 clinical trials, making it one of the largest cancer clinical trial programs in the Midwest.

Director of Clinical Research

Associate Medical Director

Interim Medical Co-Director

Interim Medical Co-Director
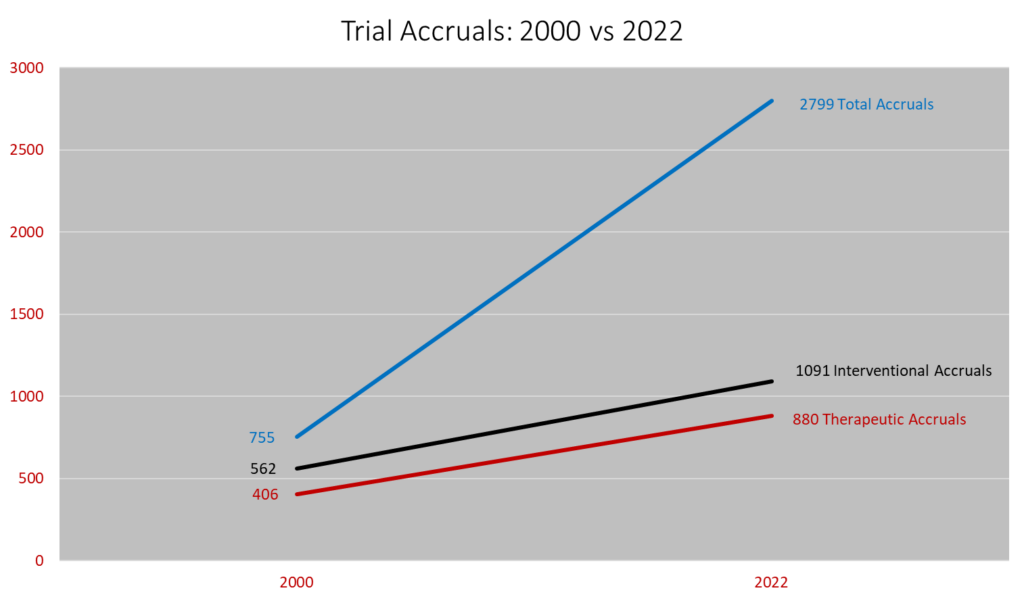
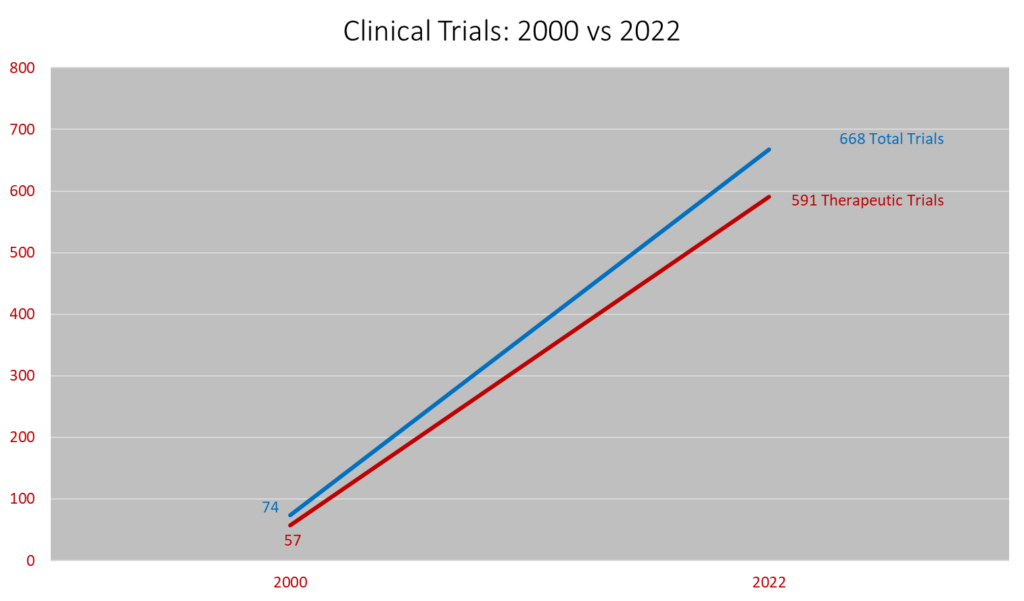
Approximately 15% of new oncology patients participate in therapeutic clinical trials.
Clinical research efforts are organized around 10 major disease-based groups:
- AIDS-related malignancies
- Breast
- Developmental Therapeutics
- Gastrointestinal
- Genitourinary
- Head and neck
- Hematologic malignancies
- Melanoma
- Neuro-oncology
- Sarcoma
- Thoracic
The Division has an extensive infrastructure to support clinical research, with 250+ professional clinical research staff, including clinical research coordinators (CRCs), research nurses, financial coordinators, regulatory coordinators, pharmacists and other support staff. The contributions of our CRCs are essential to advance our mission in developing better therapeutics for our patients. Oncology CRCs play an integral role in the conduct of high quality clinical trials; they are the link between the hypothesis and analysis. CRCs also help patients understand that their participation in clinical trials may benefit them and others in our collective fight against cancer. The primary clinical research facility is the Siteman Cancer Center on the campus of Barnes-Jewish Hospital and WashU Medicine. Research also is supported at satellite facilities at Barnes-Jewish West County Hospital, Siteman Cancer Center-South County, Barnes-Jewish St. Peters Hospital, Christian Northeast Hospital, and Siteman Cancer Center– Southern Illinois.
The Division has entered into several strategic relationships with pharmaceutical companies and contract research organizations, including Genentech, Immunotherapy Centers of Research Excellence (imCORETM) Network and the IQVIA Early Phase Oncology Network. It utilizes commercial single IRBs and operates master clinical trial agreements with more than 30 pharmaceutical and biotech companies in order to streamline operations.

Clinical research efforts by investigators within the Division have shaped standard of care treatments for cancer patients worldwide. Examples include:
- CXCR4 for mobilization of hematopoietic stem and progenitor cells (J. DiPersio, Z. Crees)
- CD20 x CD3 biospecific antibodies for the treatment of lymphoma (N. Bartlett)
- Use of next-generation sequencing to track disease progression and regression and sub-clonal architecture (T. Ley, D. Link, D. Spencer, M. Walter)
- Targeted therapy for lung adenocarcinoma with mutations in G12C KRAS (R. Govindan)
- Novel HERZ/erb2 TKI for cancer therapy (C. Ma, R. Bose)
The physicians and scientists within the Division are national and international leaders in their respective oncology subspecialties. Many serve as principal investigators for large multi-center, multi-disciplinary clinical trials and are recipients of significant NIH- and industry-sponsored grants. They work collaboratively with researchers in other departments across the medical center campus as well as with investigators in other leading oncology programs across the country and around the world. Several serve as leaders in National Cancer Institute clinical research consortiums, including the Alliance for Clinical Trials in Oncology, Blood and Marrow Transplant Clinical Trials Network, AIDS Malignancy Consortium, and the Experimental Therapeutics-Clinical Trials Network. In addition, our investigators are active members of the Multiple Myeloma Research Consortium (MMRC), Hoosier Cancer Research Network, and Sarcoma Alliance for Research through Collaboration (SARC).
- NCI Alliance U10 (PI: N. Bartlett)
U10CA180833 – Washington University/Siteman Cancer Center Lead Academic Site – N. Bartlett (Medical Oncology); D. Mutch (OBGYN), B. Siegel (Radiology), B. Kozower (Surgery), C. Robinson (Radiation Oncology)
The fundamental goal is to create a supportive environment that fosters scientific leadership and mentorship; the development of cooperative group clinical trials; substantial accrual to clinical trials across the entire NCTN; and exceptional conduct of clinical trials. - NCI PDX grant (PI: L. Ding and R. Govindan)
U54CA224083 – Washington University PDX Development and Trial Center (WU-PDTC) – R. Govindan (contact PI), Li Ding, Shunqiang Li
The WU-PDTC, as part of NCI PDXNet program, is developing and characterizing patient-derived xenografts (PDXs)across all major tumor types to advance the ability to predict clinical responses to new molecularly targeted agents under development. - NIH/NCI: 1 UG1 HL138669-01 BMT-CTN Use of memory NK cells for haploidentical allogeneic stem cell transplant (PI: I. Pusic)
The goal is to achieve designation of the Washington University Bone Marrow Transplant program as a Core Clinical Center for the BMT Clinical Trials Network (CTN).